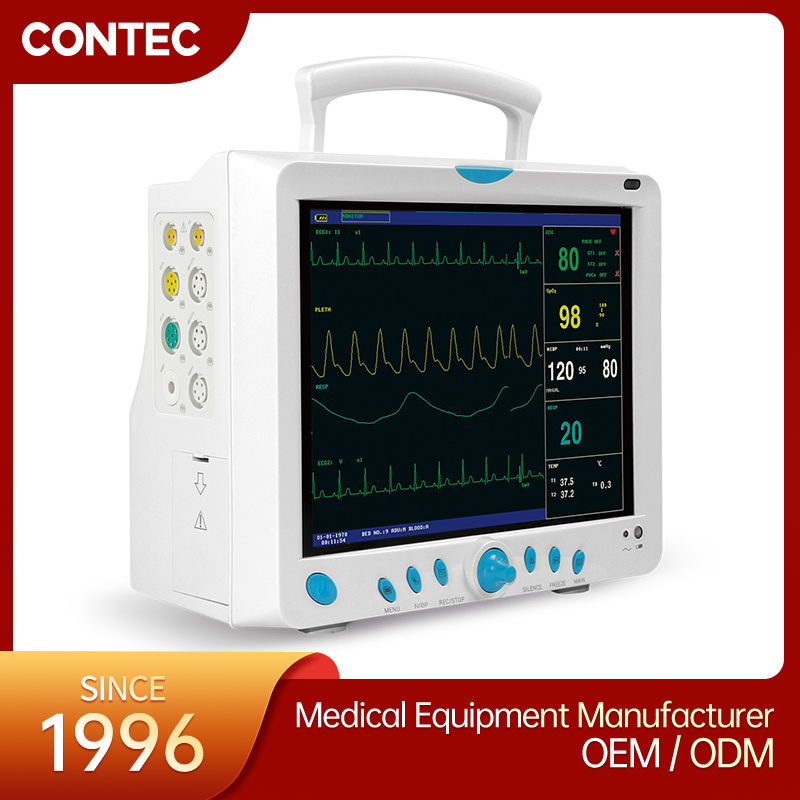
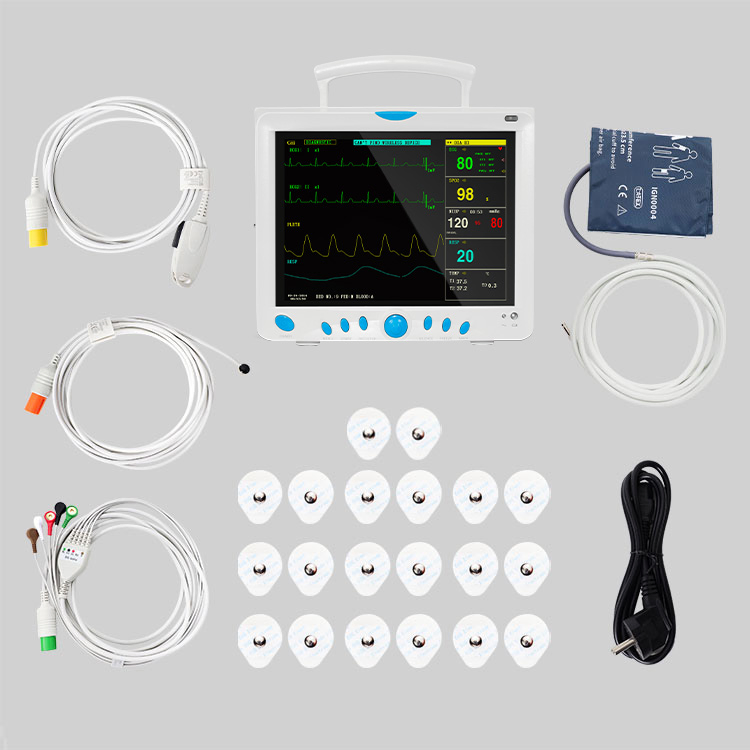
CMS9000 CE and FDA Certificated Patient Monitoring System
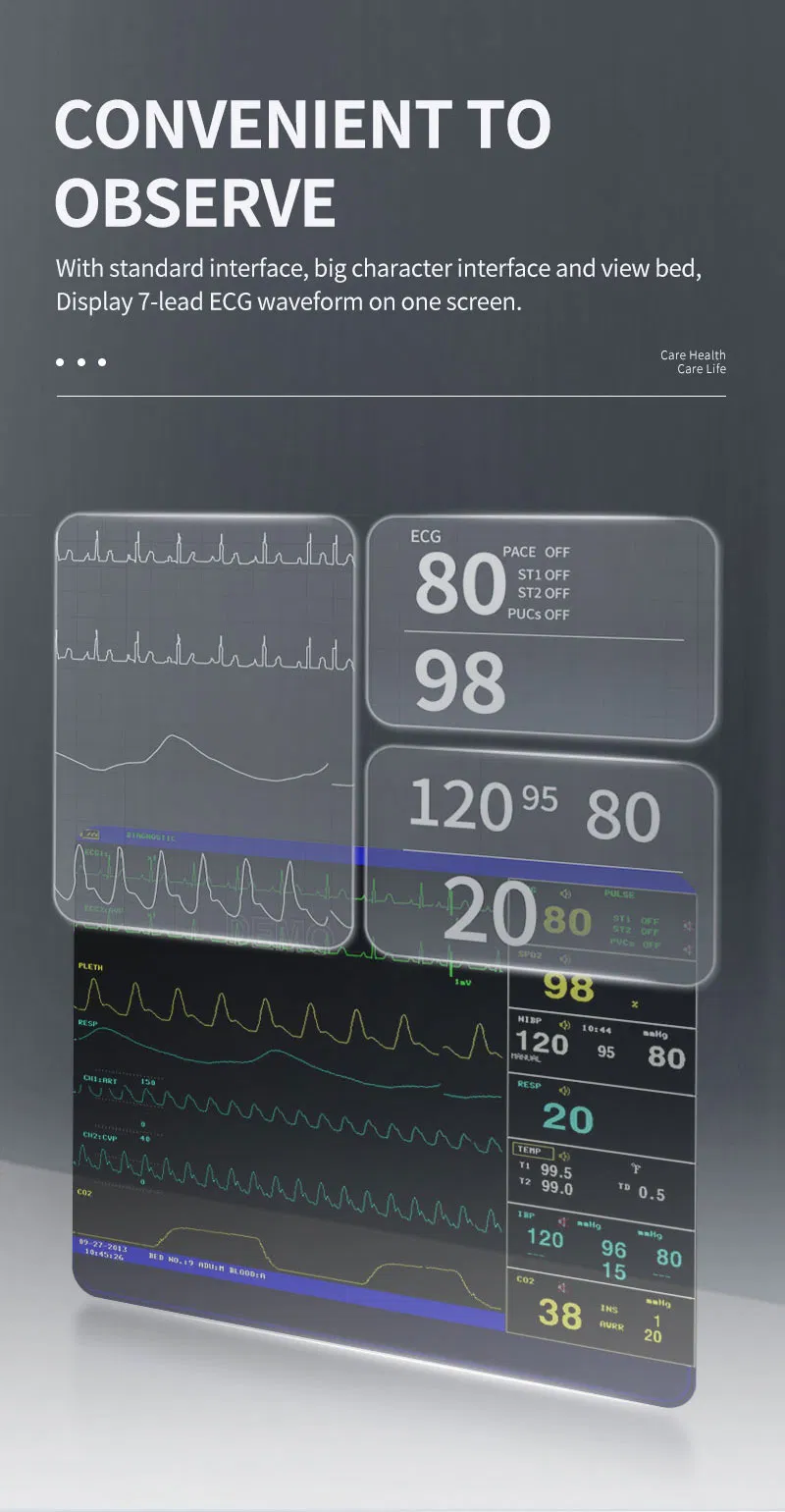
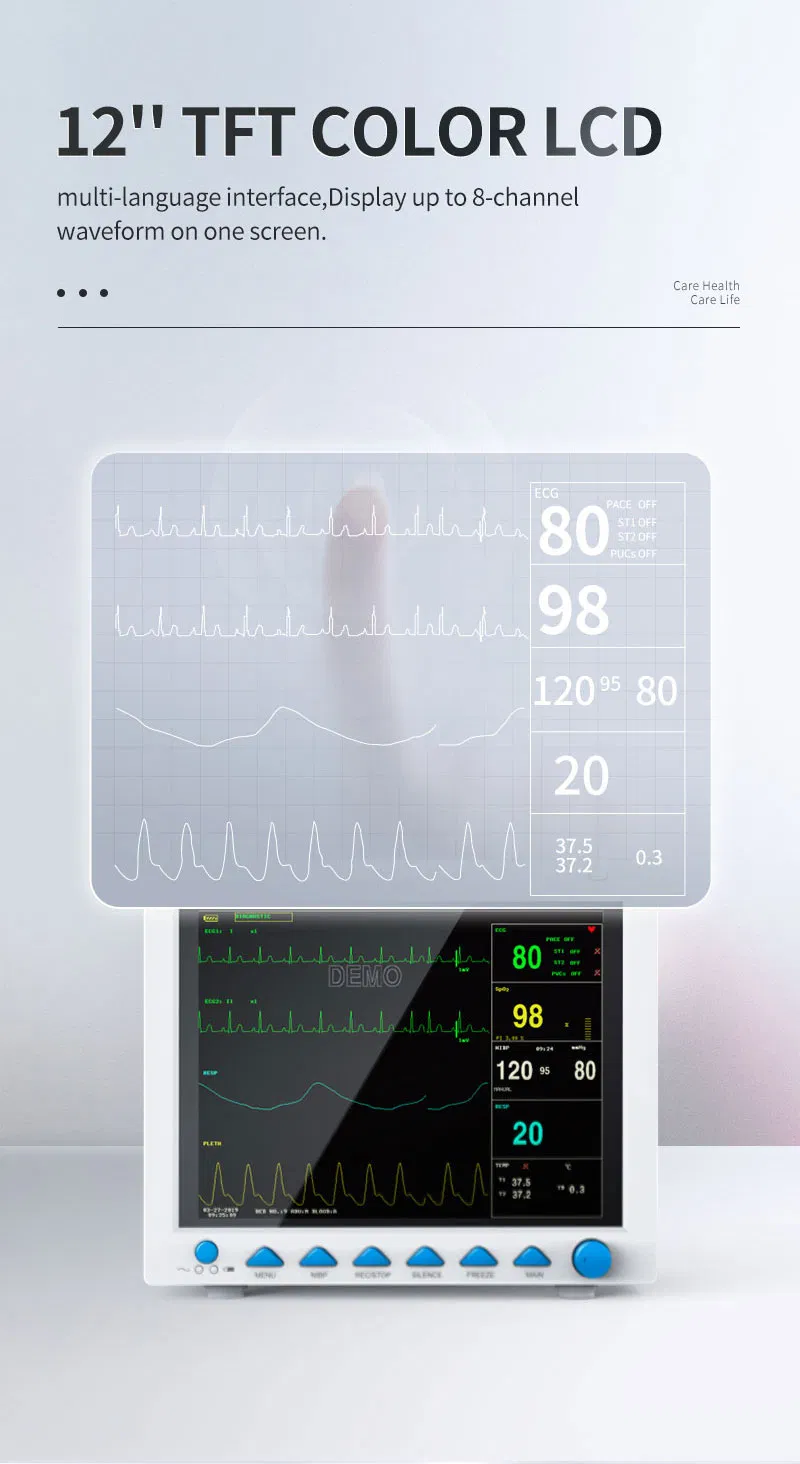
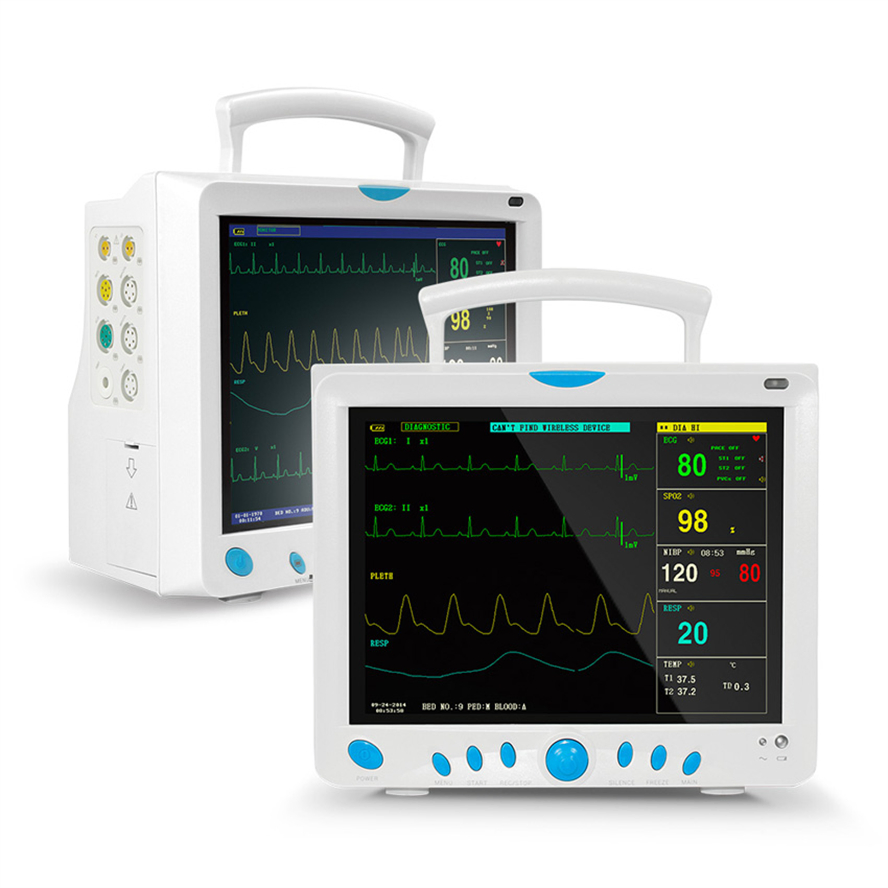
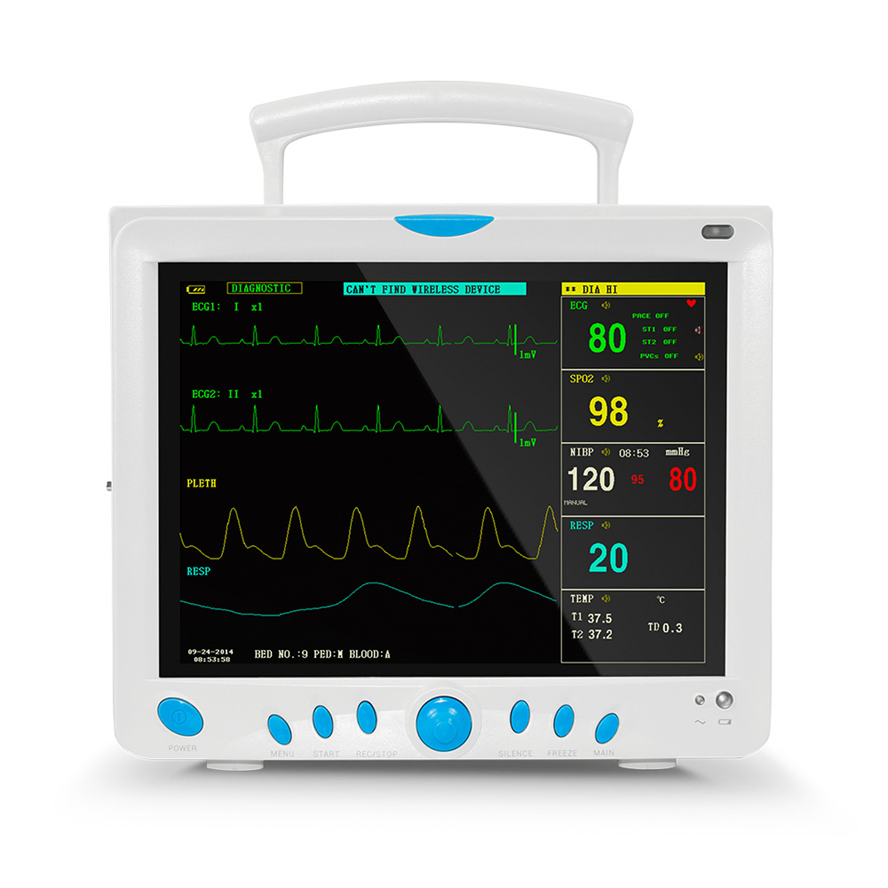
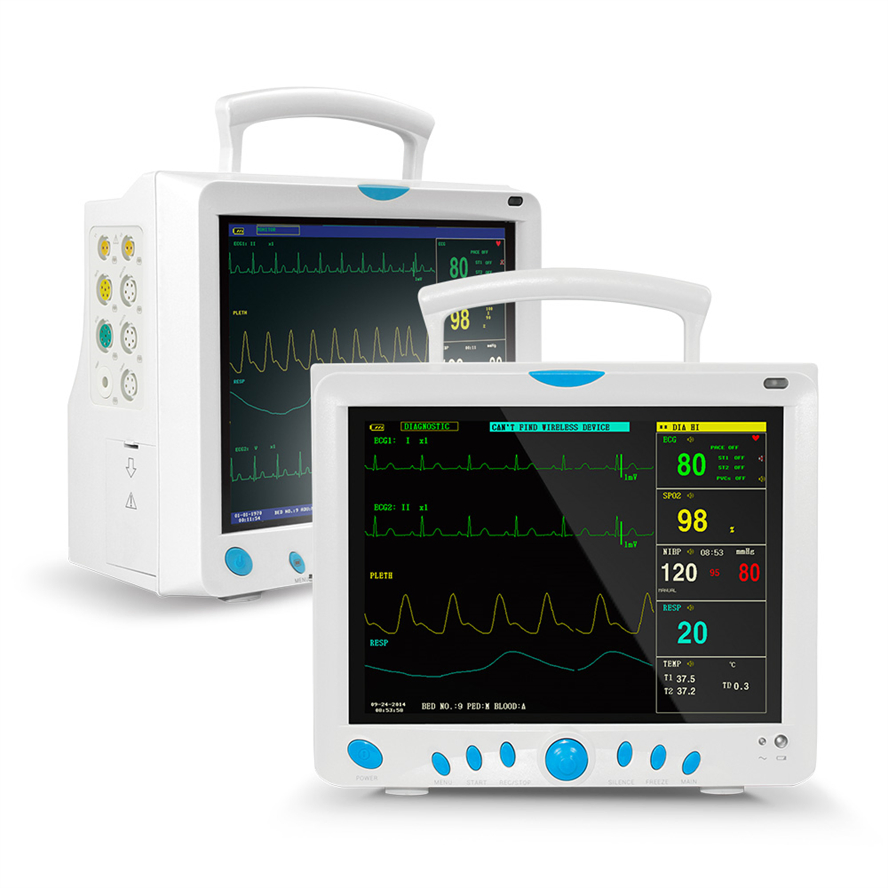
High-Res Display
12.1" Color TFT Screen, displaying up to 8 channels of waveforms simultaneously.
Portable Design
Light and portable with a built-in rechargeable battery for continuous monitoring during transport.
Advanced Analysis
Includes arrhythmia analysis, pace-maker detection, and S-T segment analysis.
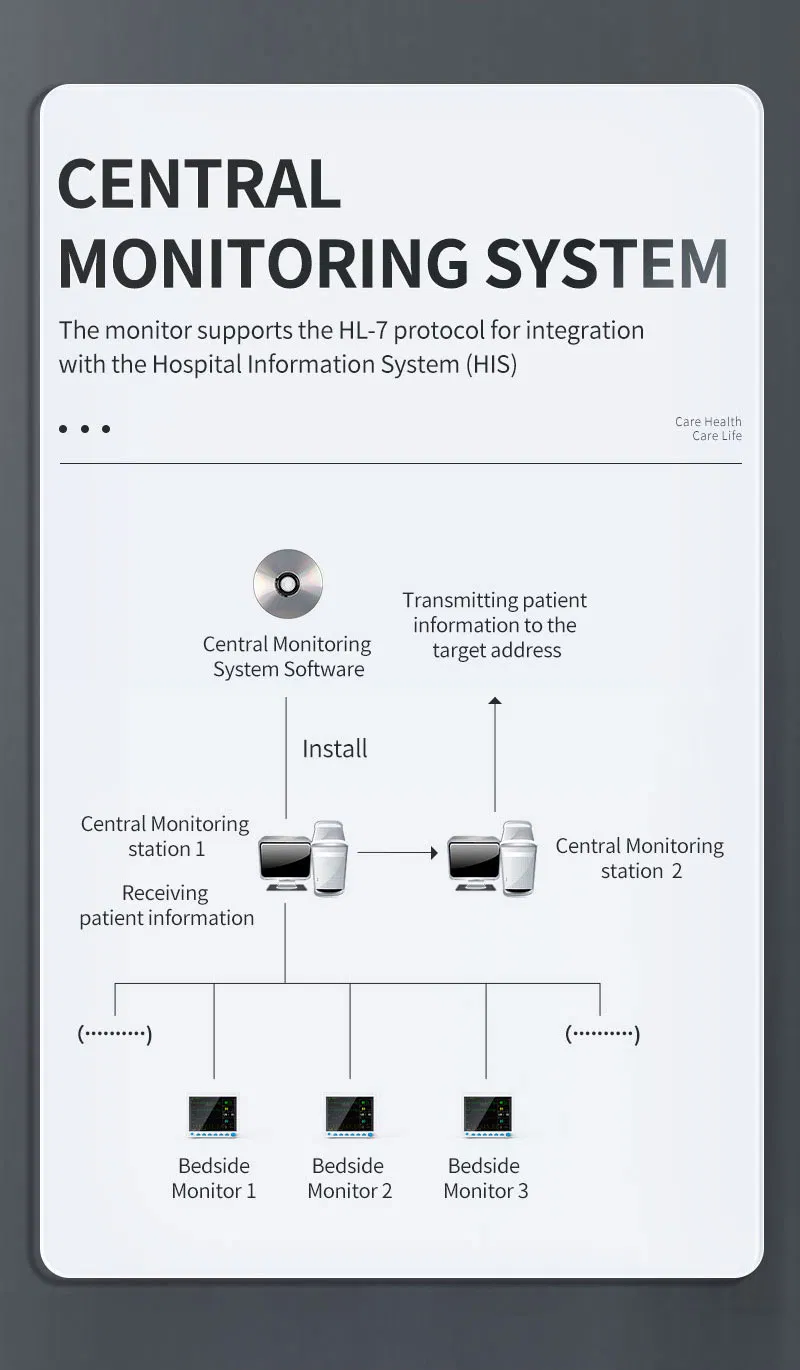
Data Storage
72-hour storage and review of trend grams and tables, plus 40-second review of holographic waveforms.
5-lead or 3-lead selectable; Lead selection: I, II, III, avR, avL, avF, V; Accuracy: ±1bpm or ±1%; Protection: Withstands 4000VAC/50Hz isolation against electrosurgical interference and defibrillation.
NIBP: Oscillometry method; Manual/Automatic/STAT modes; Range: 10-270mmHg (Adult).
SpO2: Measurement range: 0-100%; Accuracy: ±2% (70-100%).
Respiration: RA-LL impedance method; Apnea alarm included.
Temperature: Dual-channel; Resolution: 0.1°C; Range: 0-50°C.
EtCO2 (Side-stream/Mainstream), IBP (2 channels), Cardiac Output (Thermo-dilution method).
Frequently Asked Questions
What are the standard parameters of the CMS9000?
The standard configuration includes ECG, NIBP, SpO2, Respiration, Temperature, and Pulse Rate.
Is this monitor suitable for neonatal patients?
Yes, the device is designed for all-round monitoring of adult, pediatric, and neonatal patients.
Does the CMS9000 support data storage?
Yes, it provides up to 72 hours of storage and review for trend graphs and tables.
Can I add EtCO2 or IBP monitoring later?
The CMS9000 supports optional modules for EtCO2 (side-stream/mainstream), 2-channel IBP, and Thermal Printing.
Is the device protected against defibrillation?
Yes, it is electrosurgical unit and defibrillation protected, ensuring safety during surgical procedures.
What certifications does this product hold?
The CMS9000 is CE, FDA, and ISO certificated, meeting international medical standards.
 Alhilal Healthcare
Alhilal Healthcare