Alhilal Healthcare
Alhilal Healthcare
 Alhilal Healthcare
Alhilal Healthcare
In the rapidly advancing landscape of medical technology, Wireless Endoscopy Systems have emerged as a cornerstone for minimally invasive diagnostics and surgical procedures. Transitioning from traditional fiber-optic cables to advanced wireless transmission has revolutionized the clinical environment by increasing mobility, reducing the clutter of surgical theaters, and enhancing patient comfort—particularly in gastrointestinal screenings. As a leading Wireless Endoscopy Systems factory and exporter, we recognize that the integration of high-definition CMOS sensors with 2.4GHz/5GHz or ultra-wideband (UWB) wireless technology is no longer a luxury but a global standard for modern hospitals.
The global market for these systems is currently witnessing a CAGR of over 7%, driven by an aging population and the increasing prevalence of chronic digestive diseases. From Capsule Endoscopy (PillCam) that traverses the entire small bowel to high-definition wireless camera units used in ENT (Ear, Nose, and Throat) and Urology, the industrial demand is shifting toward portability and AI-assisted lesion detection. For international procurement officers, the focus has moved beyond basic visualization to data security, latency-free transmission, and long-range battery efficiency.
North America and Europe remain the largest markets, but the Asia-Pacific region is the fastest-growing hub for wireless diagnostic equipment adoption.
Integration of 4K imaging and AI algorithms for real-time polyp detection is the current benchmark for Tier-1 medical exporters.
Applications span from Gastroenterology and Urology to Orthopedics and Emergency Care, providing localized diagnostic power anywhere.
The industry is currently undergoing a paradigm shift towards "Smart Endoscopy." As a specialized factory, we are observing three critical trends that are defining the future for exporters and distributors:
1. Miniaturization and the Rise of Capsule Tech: Capsule endoscopy has moved from niche to mainstream. Modern systems now offer 360-degree coverage with dual-lens technology, allowing for thorough bowel disease diagnosis without the discomfort of traditional procedures. Our Wireless Pillcam systems are designed to offer up to 12 hours of battery life with adaptive frame rates to maximize image quality in darker regions of the digestive tract.
2. Latency-Free Wireless Transmission: The "Holy Grail" of wireless surgery is zero latency. Modern factories are now utilizing proprietary protocols that ensure 1080P and 4K video feeds reach the monitor in under 100 milliseconds, ensuring that the surgeon's hand movements are perfectly synchronized with the visual feedback.
3. Deep Learning & Diagnostic Support: High-end wireless systems are now equipped with internal AI modules that highlight suspicious areas, such as bleeding, inflammation, or abnormal vascular patterns, providing the specialist with a "second pair of eyes" during screening.
Years of Expertise
Factory Area (sqm)
Skilled Professionals
13485 Certified
As a leading Chinese manufacturer, our factory offers a unique blend of supply chain excellence, R&D agility, and cost-effectiveness that is unmatched in Western markets. The "Made in China 2025" initiative has significantly bolstered our capabilities in high-end medical electronics. When you procure from our facility, you are not just buying a product; you are accessing a robust ecosystem of medical innovation.
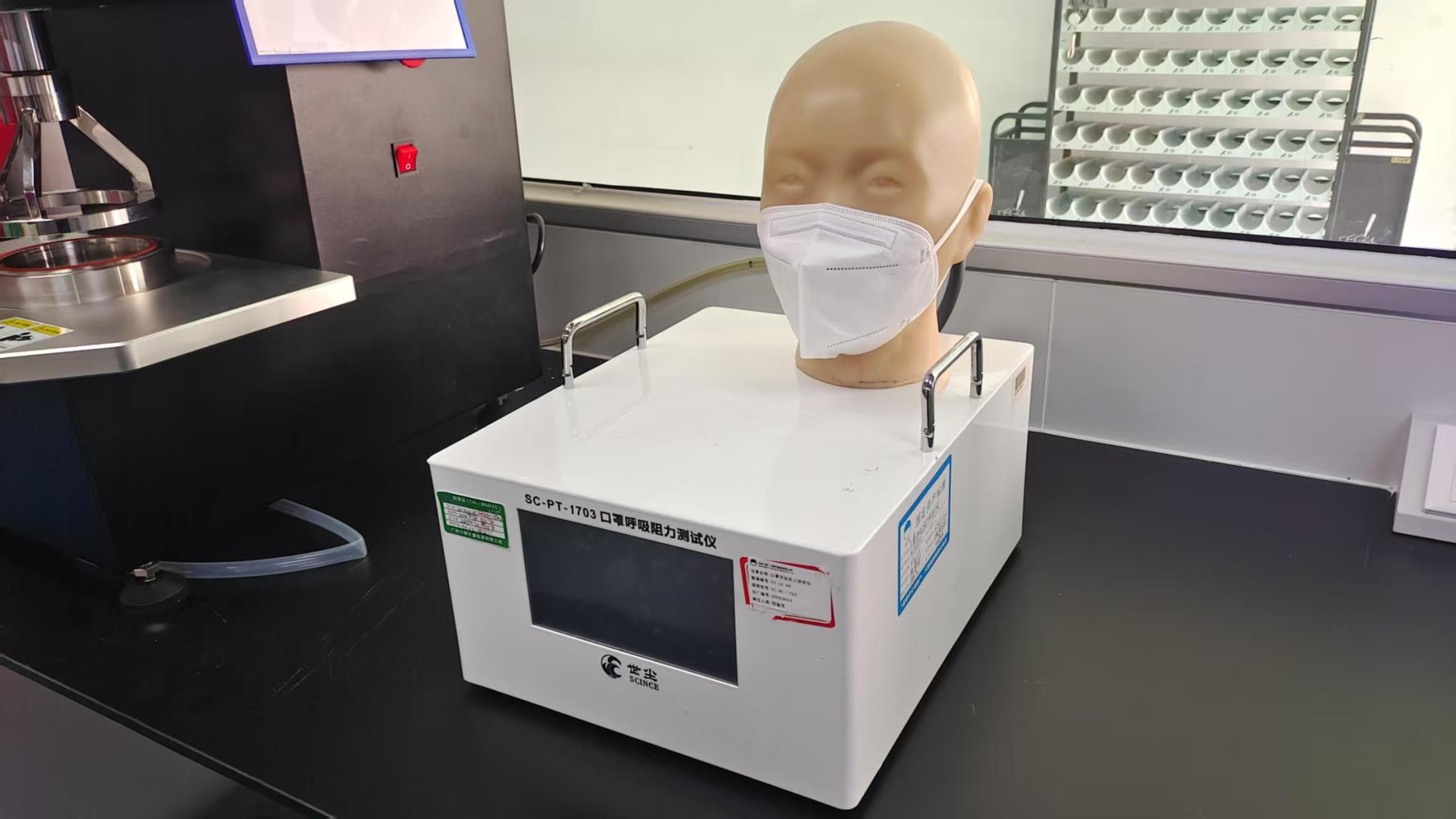
Comprehensive Quality Control: Our facility is not only focused on electronic assembly but is founded on a 15-year history of medical-grade manufacturing. With a 6,000 square meter clean workshop and rigorous ISO13485 auditing, we ensure that every endoscope meets international safety standards. Our factory utilizes advanced testing equipment, including Gas Chromatographs and Hydrostatic Pressure Testers, to ensure the durability and sterility of all associated components.
OEM/ODM Flexibility: We understand that different markets have different regulatory and language requirements. We offer localized software interfaces, custom hardware branding, and tailored imaging sensors to meet the specific needs of healthcare providers in South America, Southeast Asia, and the Middle East.






The procurement needs for wireless endoscopy differ vastly by region. In developed markets, the focus is on integration with EMR (Electronic Medical Record) systems and ultra-high-definition 4K outputs for teaching hospitals. In developing markets, the demand is for all-in-one portable solutions like our Portable Endoscopy Systems with integrated 17-inch LCD screens, which are ideal for remote clinics or emergency mobile units where external monitor setups are impractical.
Our export strategy addresses these local scenarios by providing versatile equipment such as the Electronic Uretero-Nephroscope for urology and specialized ENT systems. These devices are designed to be "plug-and-play," reducing the need for extensive training and allowing medical practitioners to focus on patient outcomes rather than technical troubleshooting.
Equipped with Flame Resistance, Blood Penetration, and Exhalation Valve testers to ensure total medical safety.
Full CE and ISO13485 certification, ensuring that our wireless systems meet the highest global regulatory bars.
Continuous investment in sensor technology and wireless protocols to maintain a competitive edge in 1080P/4K imaging.
Our factory, established in 2009, has specialized in the field of high-grade medical manufacturing for over 15 years. Today, we stand as a powerhouse of production with more than 500 dedicated employees and a sprawling facility covering more than 100,000 square meters. Our commitment to medical excellence is evidenced by our 6,000 square meter clean workshop, where we produce not only high-tech endoscopy systems but also a full range of personal protective equipment, including non-woven medical face masks, surgical gowns, and isolation suits. This diverse manufacturing capability ensures we maintain a comprehensive understanding of medical safety and hygiene, which is critical when developing invasive or diagnostic medical electronics.
For the better promising quality to our customers, our company has passed the factory audit report from ISO13485 and CE. All our products meet the high levels of quality demand and conform to worldwide standards.