



 Alhilal Healthcare
Alhilal Healthcare










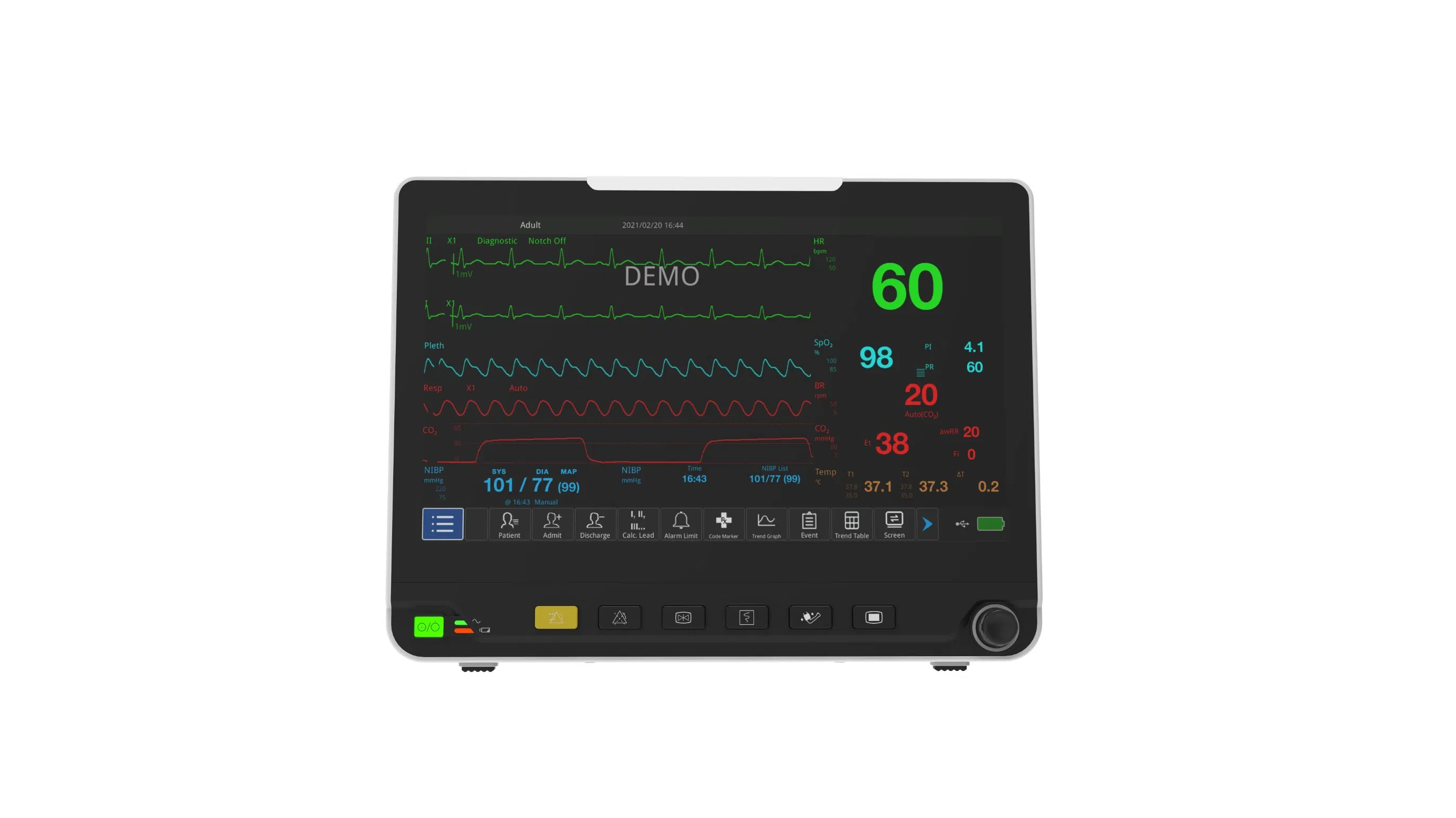
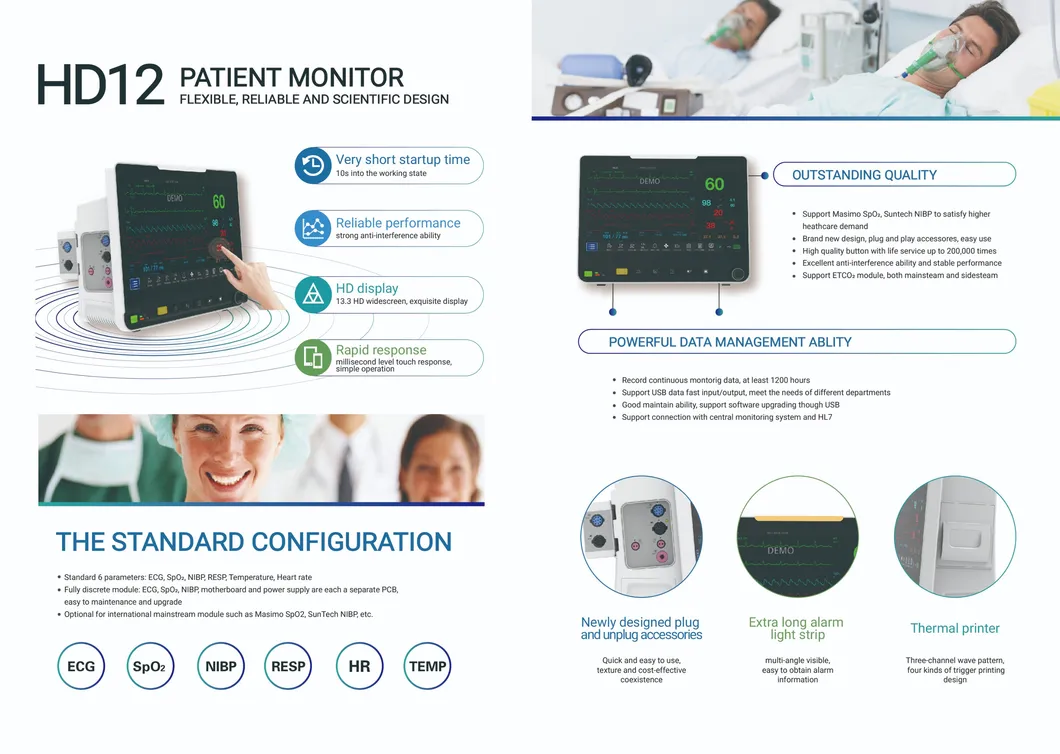
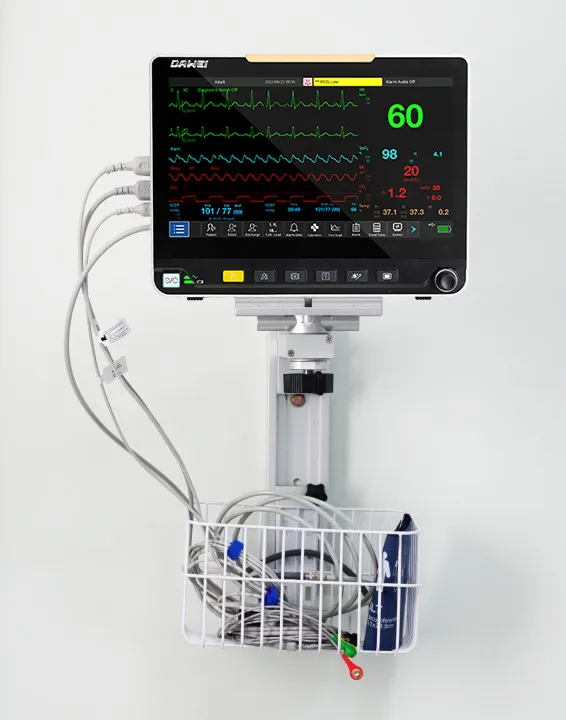
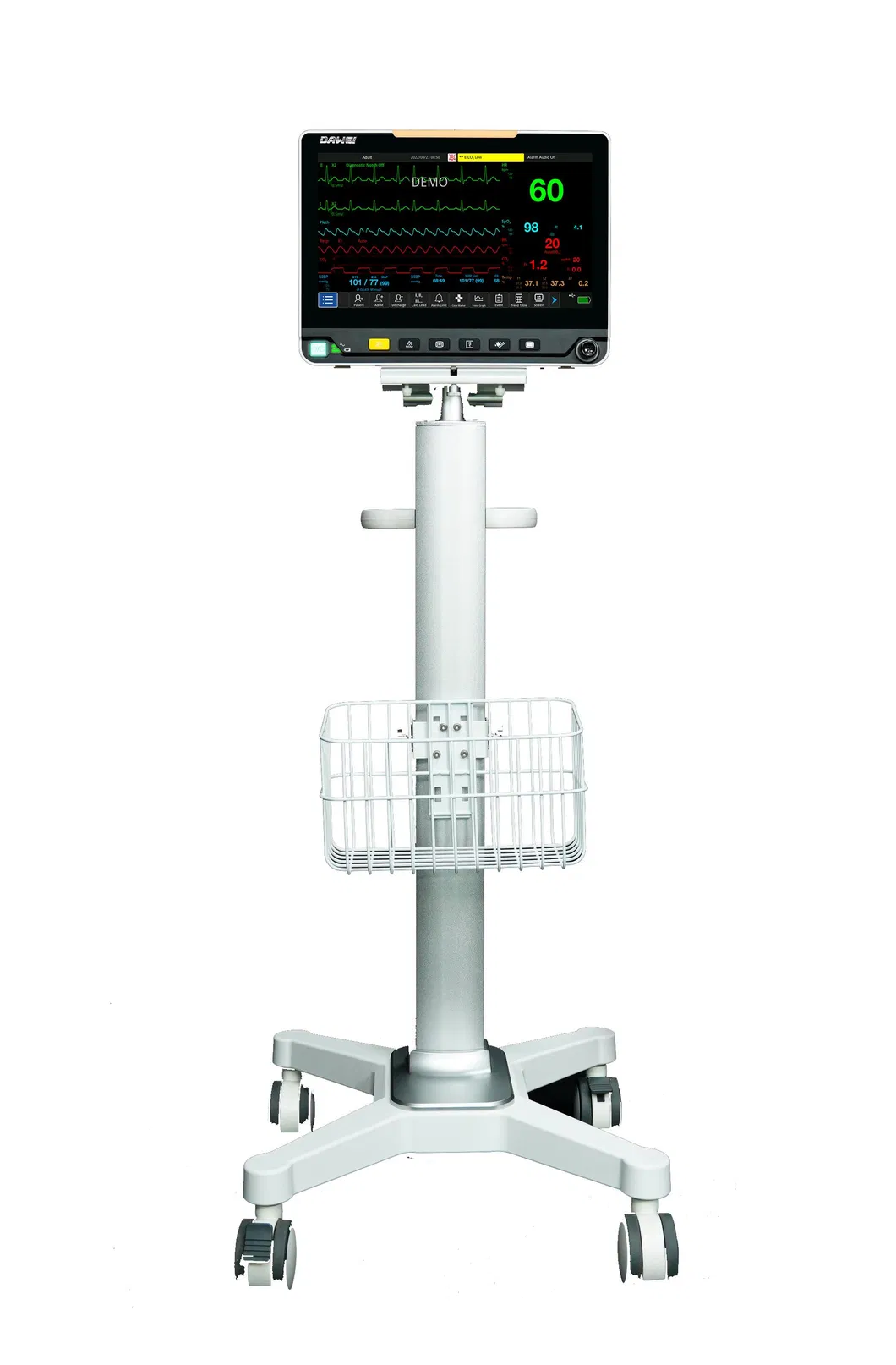
Portable Multi-Parameter monitoring solution designed for high-efficiency human-computer interaction in complex medical environments.




ISO 13485 certification is an international standard for the medical device industry worldwide. This certification emphasizes the establishment and maintenance of a quality management system, aiming to ensure that our products meet the rigorous requirements of the medical industry and consistently provide high-quality, safe, and reliable medical devices.
By complying with the ISO 13485 standard, comprehensive control is ensured from product design to production, sales, and after-sales service, meeting needs for reliability and effectiveness.
Our medical device manufacturing facility is a well-established medical device provider, recognized as a national high-tech enterprise. Dedicated to innovative development and manufacturing of ultrasound imaging, digital electrocardiograph, and patient monitoring products.
Since 2006, the focus has been on delivering professional medical solutions. The product portfolio consists of over 50 models in five major categories, including color Doppler systems, black and white ultrasound machines, digital ECG machines, and patient monitors. All systems are certified under ISO 13485 quality standards and have received necessary regulatory approvals.



Our technical experts provide comprehensive assistance and solutions. We offer remote technical support through phone, email, or remote assistance tools to assist with equipment operation, troubleshooting, and software issues. Professional training and on-site repair services are available when necessary to ensure equipment is restored to normal operation promptly.
